Rivaroxaban-discontinueringspercentages bij niet-valvulair AF in de Italiaanse praktijk
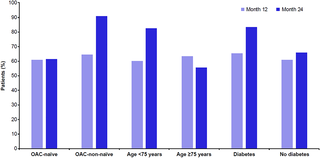
Non-adherentie aan DOAC-therapie verhoogt het risico op ischemische beroerte bij niet-valvulair AF. Circa 20% van de patiënten staakt de behandeling. Deze RITMUS-AF-studie onderzocht de discontinueringspercentages van rivaroxaban in de Italiaanse klinische praktijk.
Abstract (original)
Nonadherence to direct oral anticoagulant (DOAC) therapy exposes patients with nonvalvular atrial fibrillation (NVAF) to an increased risk of ischemic stroke and systemic embolism. Nevertheless, approximately 20% of patients discontinue treatment within the first year. In Italy, data on DOAC discontinuation rates are limited, especially in high-risk populations. RITMUS-AF, a prospective, observational cohort study conducted in 31 centers across Italy, investigated rivaroxaban treatment discontinuation in patients with NVAF in routine clinical practice. It included 805 patients aged ≥18 years with NVAF who were newly initiated on rivaroxaban. The primary endpoint was the proportion of patients who discontinued treatment during a 24-month follow-up. Secondary endpoints included the reasons for discontinuation and self-reported adherence to rivaroxaban therapy. At baseline, most patients were oral anticoagulant (OAC)-naïve (n = 599, 74.4%) and had either symptomatic (n = 364, 45.2%) or asymptomatic (n = 441, 54.8%) NVAF. The overall rate of rivaroxaban discontinuation was 8.9 per 100 patient-years (95% CI: 7.1-11.0). The main reasons for discontinuation were adverse events or physician decisions. After 24 months, high adherence was reported in 90.9% of OAC-non-naïve patients and 61.5% of OAC-naïve patients. Forty-six patients (5.7%) experienced bleeding events (with major bleeding events occurring in <0.5% of cases), and one patient (0.1%) had an ischemic stroke. In the RITMUS-AF study, rivaroxaban treatment was associated with a low treatment discontinuation rate, along with high self-reported adherence and a relatively low incidence of ischemic stroke and bleeding events in a high-risk population, findings that may help inform clinical decision-making on the use of rivaroxaban in routine practice.
Dit artikel is een samenvatting van een publicatie in PloS one. Voor het volledige artikel, alle details en referenties verwijzen wij u naar de oorspronkelijke bron.
Lees het volledige artikelDOI: 10.1371/journal.pone.0341633
