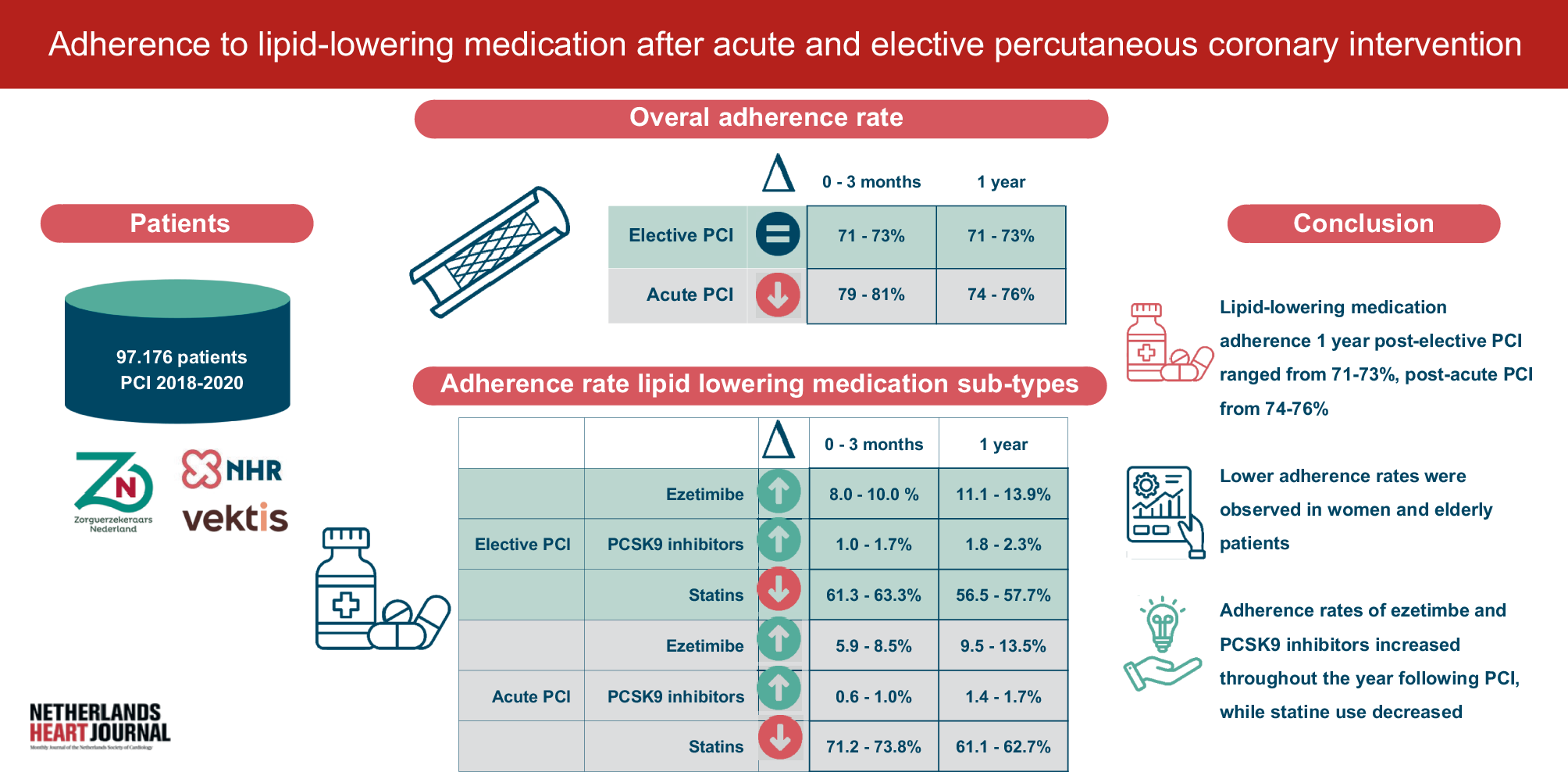
KARDINAL: tonlamarsen verlaagt angiotensinogeen maar laat geen significant bloeddrukeffect zien
In de KARDINAL fase 2-studie ontvingen 206 patiënten met ongecontroleerde hypertensie (ondanks 2-5 antihypertensiva) maandelijkse injecties met tonlamarsen, een antisense-oligonucleotide gericht op angiotensinogeen.
Na 20 weken was het angiotensinogeenniveau significant gedaald (-67% vs -23% bij placebo). De systolische bloeddruk daalde met 6,7 mmHg in beide groepen, zonder significant verschil. Het onverwacht langdurige effect van een enkele dosis bemoeilijkte de interpretatie: ook de placebogroep behield bloeddrukverlaging door de actieve inloopfase.
Simultaan gepubliceerd in JACC.
Abstract (original)
Background: Angiotensinogen (AGT), the sole substrate for renin, governs the rate-limiting step of the renin-angiotensin-aldosterone system (RAAS). Suppression of hepatic AGT synthesis represents an upstream strategy to blunt RAAS activation and lower blood pressure (BP). Tonlamarsen is an investigational GalNAc-conjugated antisense oligonucleotide that is administered once monthly as a subcutaneous injection. The KARDINAL trial is a randomized, double-blind, placebo-controlled, multicenter, phase 2 study to evaluate the safety and BP lowering efficacy of 90 mg of once-monthly tonlamarsen administration among participants with uncontrolled hypertension. Methods: The trial was conducted at 39 sites within the United States. Participants were enrolled if they were taking between 2 and 5 BP medications and office systolic BP was between 135 to 170 mm Hg. Following a placebo run-in, participants were treated for 20 weeks beginning with a 4-week single-blind active run-in (all participants administered tonlamarsen), and subsequent double-blind 1:1 randomization to placebo or ongoing monthly treatment with tonlamarsen for 16 weeks. The co-primary end points are the percent change in plasma AGT levels and the change in office systolic BP, from baseline to end of treatment, among participants randomized to ongoing tonlamarsen treatment compared with placebo at week 20. Results: 206 participants received tonlamarsen during the active run-in and 198 were randomized. At 20 weeks, angiotensinogen levels had dropped by 67.2% from baseline among those who continued taking tonlamarsen and by 23% among those who switched to placebo — a significant between-group difference of 44.1% in favor of continuous tonlamarsen. Systolic blood pressure readings dropped by 6.7 mmHg on average across both study groups, with no significant between-group difference at this time point. Conclusion: Tonlamarsen significantly reduced angiotensinogen but did not demonstrate a significant between-group difference in blood pressure reduction at week 20.
Dit artikel is een samenvatting van een publicatie in Journal of the American College of Cardiology. Voor het volledige artikel, alle details en referenties verwijzen wij u naar de oorspronkelijke bron.
Lees het volledige artikelDOI: 10.1016/j.jacc.2026.03.001