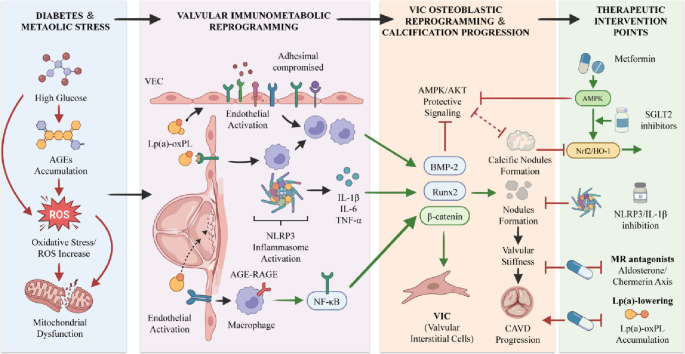
CONFIDENCE-analyse: acute eGFR-daling bij empagliflozin + finerenone is reversibel en geen reden om te stoppen
In deze prespecificeerde CONFIDENCE-analyse (n=790, T2D + CKD + albuminurie) was de acute eGFR-daling op dag 14 het grootst bij combinatietherapie (-6,6 mL/min) versus finerenone (-2,1) of empagliflozin alleen (-4,8), maar de daling was reversibel.
Het additieve UACR-verlagend effect van finerenone was niet-hemodynamisch, terwijl empagliflozin's effect deels via eGFR-verandering verloopt. De analyse ondersteunt doorbehandelen bij initiële eGFR-dip en combinatietherapie als pilaar in CKD bij T2D.
Abstract (original)
KEY POINTS: We investigated the effect of empagliflozin, finerenone, and their combination on eGFR decline in people with type 2 diabetes and albuminuria. Acute declines in eGFR occurred more in those on combination therapy, on diuretics, and with higher eGFR; eGFR changes were reversible. Finerenone's additive effect to empagliflozin on urinary albumin-to-creatinine ratio lowering is nonhemodynamic; empagliflozin lowers urinary albumin-to-creatinine ratio in part driven by eGFR change. BACKGROUND: eGFR decline is common with sodium-glucose cotransporter 2 inhibitors and renin-angiotensin system inhibitors, often prompting treatment interruption or cessation, limiting cardiorenal benefits. This mostly prespecified COmbinatioN effect of FInerenone anD EmpaglifloziN in participants with chronic kidney disease and type 2 diabetes using a UACR Endpoint (CONFIDENCE) trial analysis investigated the effect of empagliflozin, finerenone, and their combination on change from baseline in eGFR and its determinants and the relationship of change in eGFR with albuminuria reduction in people with type 2 diabetes and CKD. METHODS: Evaluable participants (N=790) with type 2 diabetes, CKD, and albuminuria, receiving stable doses of renin-angiotensin system inhibitors, were randomized 1:1:1 to empagliflozin, finerenone, or both. The primary outcome was urinary albumin-to-creatinine ratio (UACR) change from baseline to day 180. We assessed mean eGFR change from baseline at day 14 (acute), determinants of acute eGFR decline, and acute eGFR change from baseline as a mediator of UACR reduction at day 180. RESULTS: The mean acute eGFR decline was greater with combination therapy (-6.6 ml/min per 1.73 m2) than with finerenone (-2.1 ml/min per 1.73 m2) or empagliflozin (-4.8 ml/min per 1.73 m2) monotherapy; P < 0.001. Acute decline in eGFR was significantly more pronounced among participants with higher baseline eGFR and in those receiving diuretics at baseline (P < 0.001 for both factors). Baseline values for systolic BP and UACR had no statistically significant effect on acute eGFR decline. Exploratory analysis showed that acute eGFR change mediated 28% of the effect of adding empagliflozin to finerenone on UACR reduction at day 180 but only 5.2% of the effect when adding finerenone to empagliflozin. AKI was uncommon in all treatment groups. CONCLUSIONS: Acute eGFR decline was significantly associated with combination therapy, higher baseline eGFR, and diuretic use at baseline. CLINICAL TRIAL REGISTRY NAME AND REGISTRATION NUMBER: ClinicalTrials.gov, NCT05254002.
Dit artikel is een samenvatting van een publicatie in Journal of the American Society of Nephrology : JASN. Voor het volledige artikel, alle details en referenties verwijzen wij u naar de oorspronkelijke bron.
Lees het volledige artikelDOI: 10.1681/ASN.0000001071
Lid worden van HartVaat.nl?
Gratis — en we stemmen het nieuws en de literatuur af op uw vakgebied.